I’m back from an intense semester of learning and teaching Developmental Biology. One theme that emerged from my studies was that the development of organisms is centered around gene expression and cell to cell signaling. Often times, one cell will differentiate into its mature form, and then release a signaling protein that tells neighboring cells what to develop into. For instance, the nervous system is induced by signals released from the embryonic backbone. There are a number of common signals that are used over and over throughout development, like BMP, Wnt and Shh.
A recent paper by Sanders et al., published in Nature, looked at how distant cells can signal to each other via the Shh pathway. Unfortunately for Developmental Biology teachers everywhere, Shh stands for Sonic Hedgehog. Oftentimes, strange or humorous gene names like this can be blamed on the fruit fly researchers who first discovered the gene, but in this case everyone is to blame. This gene was originally discovered by researchers studying fruit fly embryonic development; they named the gene hedgehog because the mutant embryos had lots of tiny bristles all over, kind of like a hedgehog. The mammalian researchers took it to the next ridiculous level, by naming the mammalian version of this gene Sonic Hedgehog. The Shh protein is a secreted signal that binds receptors on other cells, which activate gene expression in the receiving cell. Shh signaling is important for specifying many different cell fates, such as the different neurons in the spinal cord, the cells that become the vertebrae, as well as the formation of the digits of the hand.
Although Shh is secreted from the cell, it has chemical modifications that make it stick to the plasma membrane that surrounds the cell that released Shh. How then can Shh induce the development of cells that are located at a distance? Well, the answer is by stretching out long cellular extensions with Shh localized at the tip.
Shh Filopodia
Sanders et al. did live imaging of cells in the developing limb of the chicken using fluorescent proteins. They did some genetic trickery so only a few cells were labeled in red and others in green. This way they could detect individual cells in a sea of unlabeled cells and examine their structure in real time. They observed individual cells extending long protrusions, called filopodia, from the cell bodies. These filopodia could stretch long distances (150 micrometers, like 3-5 cell widths) and were dynamic– retracting and growing over time.
How to think about filopodia? Imagine a stretchy balloon with a stick inside of it. If you could push that stick into the wall of the balloon, the balloon would protrude from that one spot as the stick pushes it out. That is like a filopodia, where the balloon wall is the plasma membrane and the stick is a protein called Actin. Actin forms long chains that can grow, pushing out the membrane in front of it.
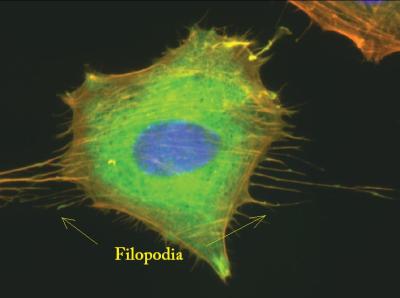 |
| The thin, string-like extensions from this cell are filopodia and are filled with Actin. Image from proteopedia.org |
The authors then labeled the Shh protein with another fluorescent marker and saw that it localized to the tips of filopodia. Not only that, but the filopodia expressing Shh were more stable and did not retract as often. In order for Shh to act as a signaling molecule, it has to bind a receptor on another cell. Using a different color, the authors observed two co-receptors for Shh localized to filopodia from other cells. They even saw filopodia from two different cells make contact with each other, where one cell expressed Shh and the other expressed the receptors.
This is amazing! Instead of releasing a signal out of the cell with the hope that it goes to the right place and isn’t degraded, the cells literally grow to the right place with the signal on their membranes. This is like hand delivering a note to your coworker, rather than making the note into a paper airplane and throwing it in the direction of their desk.
A study like this could not have been done before recent innovations in live imaging and molecular biology to introduce the fluorescent proteins into the cells. The filopodia are not preserved during the more traditional, static method of fixing cells with formaldehyde and then staining them. Who knows what other tricks live cells use during embryonic development. I suspect this is only the beginning.
